It can explode when mixture with air and at high temperatures. Health effects / safety hazards: Hydrogen is harmful to human health. Hydrogen can also be used to produce batteries or to produce energy through the nuclear fusion power plants. It is also used in the production of petroleum oil. Uses: Hydrogen can be used as a reactant in the production of ammonia and some acids as HCl and H2S.  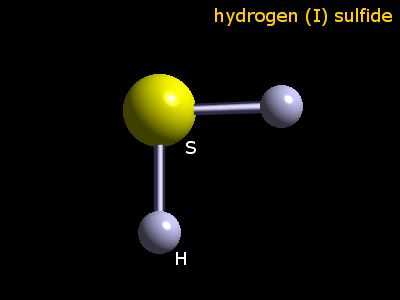
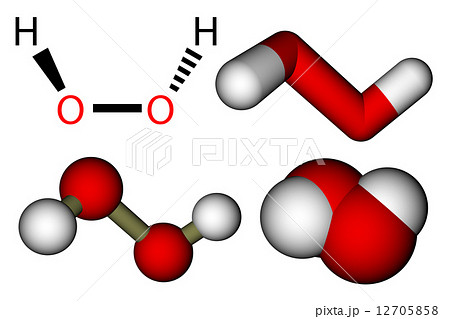
Hydrogen gas can react with other elemental compounds as halogens to forms hydrides and acids: Growing evidence has shown that hydrogen gas can either alleviate the side effects caused by conventional chemotherapeutics. In general, it is considered very reactive because the mixtures with air can be explosive, but only at temperature up to 500 ☌. Recently, hydrogen gas (formula: H 2) emerges as another GSM which possesses multiple bioactivities, including anti-inflammation, anti-reactive oxygen species, and anti-cancer. Its melting point is -259.16 ☌ and its boiling point is -252 ☌.Ĭhemical properties: Hydrogen gas is made of two molecules of elemental hydrogen H. Physical properties: Hydrogen is a colorless gas. The compound hydrogen chloride has the chemical formula H Cl and as such is a hydrogen halide.At room temperature, it is a colourless gas, which forms white fumes of hydrochloric acid upon contact with atmospheric water vapor.Hydrogen chloride gas and hydrochloric acid are important in technology and industry. When produced by micro-organisms, the H2 is biosynthesized by the action of enzymes called hydrogenases. It can be produced by the electrolysis of water resulting in molecular oxygen and hydrogen: Preparation: Hydrogen is produced by some methods: It can also be produced by some micro-organisms. Occurrence: Hydrogen is the most abundant gas in the universe, however in the Earth, is found in traces in the atmosphere and in some planets and stars as Jupiter and the Sun. Its chemical structure can be written as below, in the common representations used for organic molecules. The molecule is formed by two hydrogen atoms joined by one ionic bond. The molar mass depends on the hydrogen isotope you are considering the hydrogen (formed by a two nuclei with one proton each one) has a molar mass of 2,00 g/mol the deuterium (formed by a two nuclei with one proton and one neutron each one) has a mass of 4,00 g/mol and the tritium (formed by a two nuclei with one proton and two neutron each one) has a mass of 6,00 g/mL. Formula and structure: The hydrogen chemical formula is H 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
